We know quite a bit about how lupus develops, but since the causes vary from person to person, we do not fully understand how different factors contribute to disease in different people. The first description of lupus goes all the way back to 400BC. Since then, various factors contributing to the disease have been identified including genetic susceptibility, environmental triggers, and sex. So, what is our current understanding and what are some of the latest developments?
What is lupus?
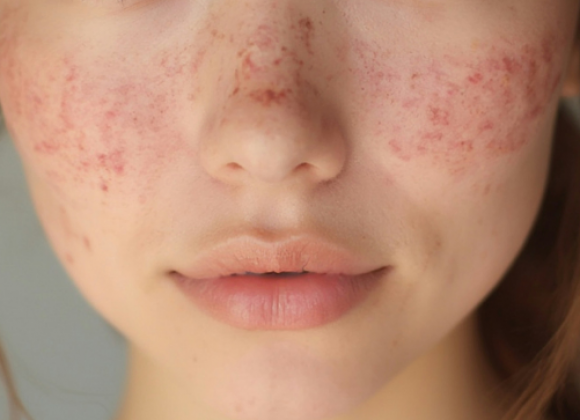
When people say ‘lupus,’ they likely are referring to systemic lupus erythematosus, or SLE, the most common type of lupus. Other types include cutaneous lupus, which is focused on the skin, drug-induced lupus which is temporarily induced from medications, and neonatal lupus affecting newborns. While SLE is more prevalent in women and people from historically marginalized communities, it affects people of all genders and ethnicities.
This disease is mainly driven by B-cells, special immune cells that produce antibodies. These are proteins that detect and stick to germs, flagging them for destruction by the immune system. B-cells can remember prior encounters with germs and quickly respond when they reencounter them later.
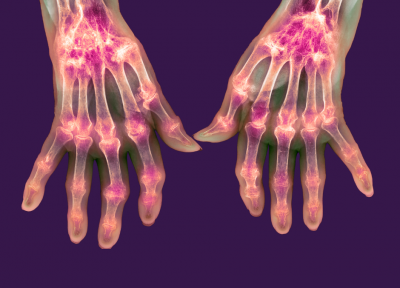
In lupus, the body mistakenly makes antibodies that attack itself, called autoantibodies. This leads to chronic inflammation as the body continuously attacks itself. In lupus, the immune system can mistakenly attack healthy tissue throughout the body, affecting major organs including skin, brain, heart, joints, nerves, blood, kidneys, and/or lungs. While treatments are available to address the symptoms, they do not cure the disease, and some patients’ immune system fails to respond to currently available drugs. Lupus research is crucial as it aims to address these issues.
Resetting the Immune System: CAR-T Cells
Chimeric Antigen Receptor T-cell (CAR-T) therapies were originally developed as a cancer treatment, but researchers are studying how to use them to target the B-cells that drive SLE. Rather than broadly suppressing the immune system, like current treatments do, CAR-T cells can eliminate B- cells. In this approach, doctors collect a patient’s T-cells and genetically engineer them so they can recognize and destroy autoantibody producing B-cells which drive lupus inflammation. The depletion of B-cells by CAR T-cells can provide a ‘reset’ for the immune system. In clinical trials, some patients have achieved long-lasting remission without the need for additional medication. The idea is that by removing malfunctioning immune cells, the body might be able to re-build a healthier immune system from scratch.
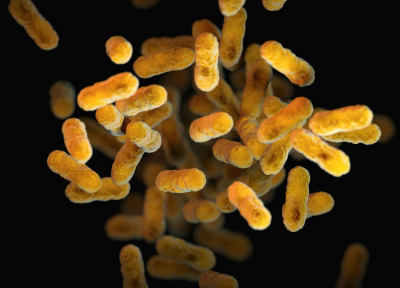
The missing link? Researchers Connect EBV to Lupus
About 95% of adults will be infected with Epstein-Barr Virus (EBV) at some point their life. After infection with EBV, the virus stays around in the body in an inactive state and for most people their immune system is able to keep EBV in-check. Scientists at Stanford University recently linked this common virus to lupus, with lupus patients having roughly 25-times more EBV-infected B-cells than healthy individuals. EBV seems to turn on pro-inflammatory B-cell genes which leads to more autoantibodies and can lead to lupus.
If almost everyone has been exposed to EBV, then why don’t more people have lupus? The researchers think that EBV’s effect on B cells may be strain-specific, with only certain strains of EBV triggering lupus in people at risk due to their genetics. Some current therapies for lupus involve removing B-cells from the body, but that is rarely 100% effective. Researchers are looking at whether replacing EBV-infected B-cells in lupus patients with non-EBV infected B-cells helps patients. There is also a clinical trial for an EBV vaccine which could prevent lupus development.
The Future? Predicting Lupus Before it Starts
Another promising direction of research focuses on detecting lupus earlier–possibly even prior to being symptomatic. Using advanced tools like genetic analysis, immune profiling, and computational models, scientists are beginning to identify patterns that may predict who is at risk for developing lupus.
Researchers have discovered that certain immune changes, including the appearance of autoantibodies, can occur years before a patient is officially diagnosed. By studying blood samples and genetic data from large patient populations, scientists are beginning to map these early warning signs. In the future, these insights could allow doctors to identify individuals at high risk before significant organ damage occurs. The shift toward earlier detection reflects a broader movement in medicine toward treating disease before it has a chance to fully take hold.
Continual discoveries are transforming how researchers think about the disease–myself included. New discoveries suggest that we may finally move beyond symptom management and toward prevention, long-term remission, and hopefully a cure.
Michelle Goldberg is earning her PhD in biochemistry under Dr. Lee Ann Garrett-Sinha at the University at Buffalo, SUNY.